PharmaGuard®
Blister Packaging


A growing number of companies in the pharmaceutical industry are anchoring sustainability aspects in their strategic corporate goals – and sending important signals to the market. In doing so, they are taking steps to reduce their ecological footprint and ultimately achieve climate neutrality. One catalyst on the road to a more sustainable future is packaging. Or rather, the substitution of packaging concepts that are not particularly environmentally friendly with recyclable alternatives.
We focus on polypropylene-based packaging concepts in the area of blister packaging for solid pharmaceutical products or nutraceuticals.
These solutions are classified as recyclable and meet current sustainability requirements. Thanks to our expertise as well as many years of project-based experience, we can actively help our customers to convert their existing packaging solutions – and offer you the best possible alternative, tailored to your unique requirements.
Ultimately, companies that are already taking steps to tackle the issue of sustainability and responsible packaging today will also be optimally positioned to meet future challenges – and will actively help to increase sustainability with regard to the environment, industry, and society.
So let’s take a huge step towards greater sustainability together! After all, our future depends on it.
Click here to download whitepaper


As a PP-based mono-material solution, our innovative blister film is recyclable – and meets the special requirements of the pharmaceutical industry. The high-performance top and bottom webs PharmaGuard are manufactured using a unique coextrusion process. They offer optimum barrier properties, are characterized by good processability on existing packaging lines, and offer a high degree of flexibility thanks to a wide range of thicknesses.
Compared to the other PP-based blister film solutions on the market, our blister concept also stands out due to its very high transparency and is ideal for packaging medications such as tablets and other products.
PharmaGuard convinces customers thanks to its highly favorable eco-balance. A screening life cycle assessment conducted by Sphera has proven that it is associated with a reduced climate impact (in CO2-eq) as well as lower energy and water consumption compared to other popular blister solutions.
All this contributes to the environmental impact of packaging materials.
PharmaGuard can be fed into mechanical recycling as a complete mono-material solution. This means a future-proof solution is already available today – which will also meet the legal requirements of tomorrow.

Depending on the product to be packaged, our PharmaGuard films can be equipped with different barrier and mechanical properties for optimum product protection and maximum stability.
The broad range of applications extends from packaging medications such as tablets to other products.
Another feature that sets our films apart from the competition is their excellent pharmacopeia-compliant water-vapor-barrier properties over the entire residual wall thickness. Since the barrier effect in polypropylene is created by the material itself and not by means of a comparatively thin coating, the measurement of the residual wall thickness provides more reliable information about the expected barrier effect. This also facilitates quality control in the packaging process.
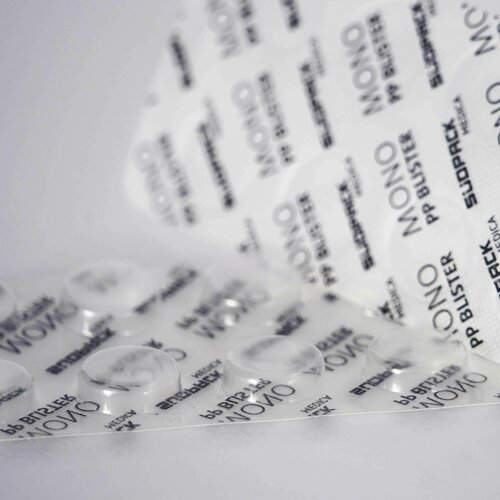
The recyclable blister solution PharmaGuard offers thanks to its large processing window, excellent thermoforming performance and uniform demolding behavior. The low, yet reproducible shrinkage behavior results in high dimensional stability, which in turn guarantees a smooth packaging process, even on standard packaging machines. High reproducibility creates comparable thermoforming and sealing conditions both within each delivery batch and from batch to batch – a prerequisite for reliable processes and packaging.

At SÜDPACK, we are specialized in creating high-performance films and packaging solutions that are based on a highly efficient use of materials. Compared to conventional packaging concepts, our products consume significantly fewer resources while maintaining high yields. Our innovative PharmaGuard for example, also convinces with excellent material efficiency.

How can a collaboration between a packaging manufacturer and a machine builder advance the sustainability of packaging?
Our CEO, Thomas Freis, and IMA's Head of Sales, Thomas Fricke, provide exciting insights in the following interview.

You can have the recyclability of your overall packaging solution evaluated by an external, independent institute. The necessary information about our films can be found in our product specifications. We would be happy to assist you in this matter.
During production of composite or multilayer films, the individual films are first extruded, then printed as needed and, depending on the application, laminated to form a high-performance composite.
As a rule, composites of this kind are based on different material structures that currently cannot be fed to a material stream. Single-material films, or mono-materials, on the other hand, are films in which the majority of the material components belong to the same family of polymers, so the films can be fed into an existing material stream for mechanical recycling.
These monomaterials contribute to improving the recyclability of packaging, especially blister packaging, and reducing the CO2 footprint.
SÜDPACK high-performance films are developed according to the "design-for-recycling" specifications. However, with regard to the recyclability of the resulting packaging, there is no simple answer to this question.
The current legislation stipulates that the unfilled packaging as a whole must be included when calculating the recyclability of packaging – this includes all the corresponding packaging components, such as labels, sealing films and closures.
However, since SÜDPACK is solely the manufacturer of packaging films that are processed by our customers into final packaging, we do not always have access to the required information regarding the final packaging and its use. The recyclability calculation should therefore be performed by the manufacturer of the final packaging.